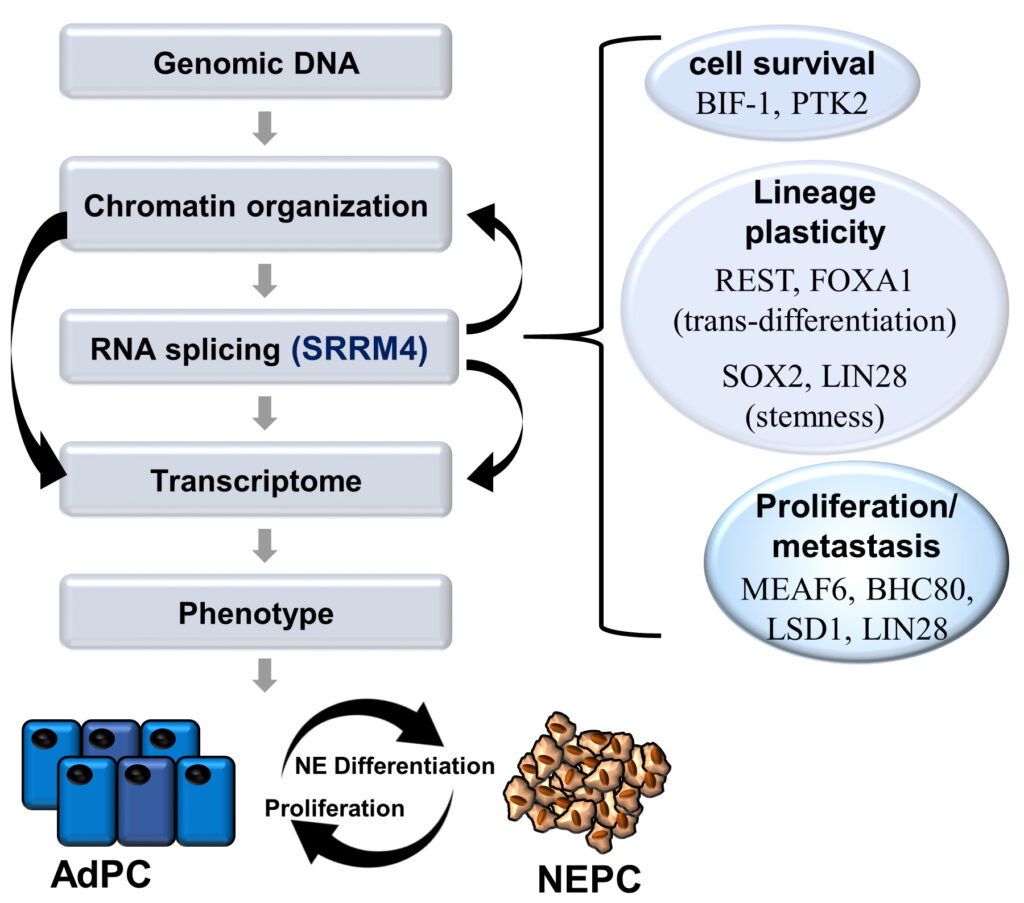
We investigate how RNA splicing processes re-program prostate cancer cell phenotypes from luminal epithelium lineage to neuroendocrine lineage. Specifically, the RNA splicing SRRM4 targets microexons from various epigenetic modifiers, transcription factors, and kinases to transform prostate adenocarcinoma into neuroendocrine prostate cancers.
________________________________________
We apply computer-aided drug design (CADD) to develop small molecule inhibitors targeting DNA topoisomerase II and Lin28 to block their interactions with nucleotides.
________________________________________
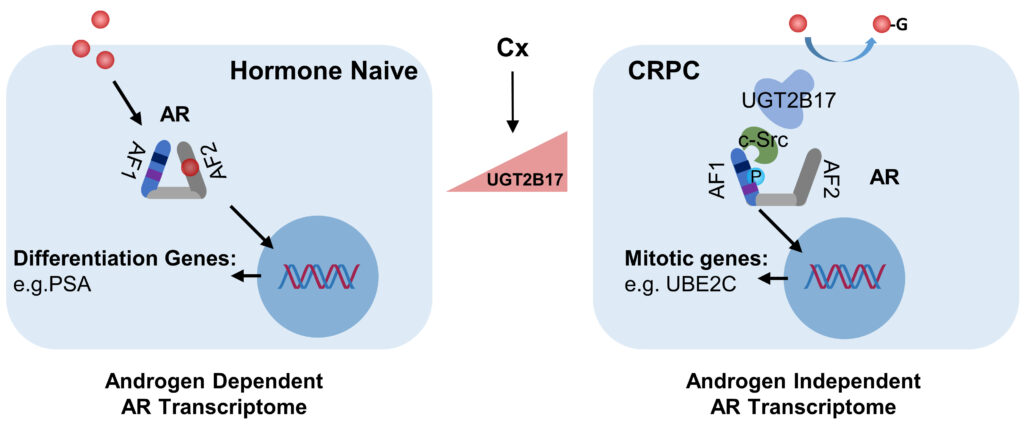
We investigate how AR signaling is transformed from being ligand-dependent to ligand-independent during CRPC progression. One of the determining factors is UGT2B17, which exerts multiple non-enzymatic activities to activate AR in the absence of androgens.
________________________________________
We apply various molecular techniques on tissue microarrays to study protein and RNA expression, protein-protein interaction, RNA splicing, and genomic DNA mutations. These tools allow us to establish molecular changes in association with disease progression, tumor cell morphology, and lineages.